The (1−x)MgO–xLiF ceramics (x = 0.02–0.08) were successfully sintered when the ceramics were sintered at 950 °C for 4 h in covered crucible. From the crystal structure analysis, it was found that a small amount of Li+ cation occupied Mg2+ site in MgO ceramic; the formation of oxygen vacancy induced by Li substitution for Mg was suggested by the evaluation of the bulk conductivity and the calculation of density of state (DOS) for the (Mg13O43)−60 and (Mg11Li2O42)−58 cluster models. As for the microwave dielectric properties of the (1−x)MgO–xLiF ceramics, the dielectric constant εr and the temperature coefficient of resonant frequency values of the ceramic were independent of the lithium fluoride (LiF) content, and these values were approximately 9.5 and −62 ppm/°C. On the other hand, the quality factor 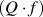 values strongly depended on the LiF content. As a result, the highest
values strongly depended on the LiF content. As a result, the highest 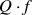 value of 282,230 GHz was obtained at x = 0.04. From these results, it is determined that the LiF addition is effective in reducing the sintering temperature of MgO without any detrimental effect on the microwave dielectric properties of MgO ceramics.
value of 282,230 GHz was obtained at x = 0.04. From these results, it is determined that the LiF addition is effective in reducing the sintering temperature of MgO without any detrimental effect on the microwave dielectric properties of MgO ceramics.



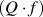 values strongly depended on the LiF content. As a result, the highest
values strongly depended on the LiF content. As a result, the highest 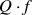 value of 282,230 GHz was obtained at
value of 282,230 GHz was obtained at 